Polaire en apolaire moleculen Tussentijdse toets Downloadbaar lesmateriaal KlasCement

Chemie polair en apolair Olie azijn die elkaar eigenlijk afstoten. Deze vloeistoffen kan je
Moleculen die een verschil in lading hebben noemen we polaire moleculen. Moleculen die geen verschil in lading hebben noemen we apolaire moleculen. De lading in een polair molecuul geven we aan met de griekse letter delta 𝛿 en een + of - teken. Moleculen die een polair zijn hebben een dipoolmoment en ze kunnen een dipool-binding aangaan.

PPT 2.Fijnstructuur van moleculen 2.2 Soorten bindingsmodellen PowerPoint Presentation ID
A solvent is simply a substance that can dissolve other molecules and compounds, which are known as solutes. A homogeneous mixture of solvent and solute is called a solution, and much of life's chemistry takes place in aqueous solutions, or solutions with water as the solvent. Because of its polarity and ability to form hydrogen bonds, water.
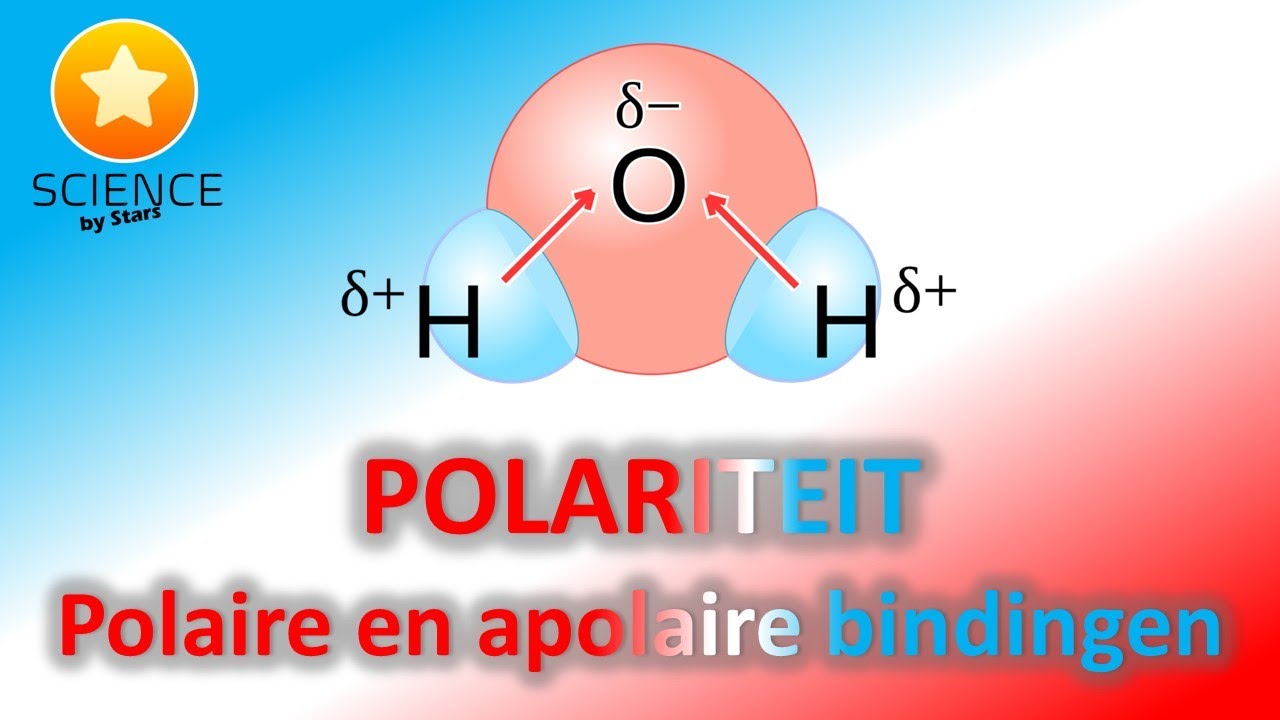
Polariteit Polaire en Apolaire binding YouTube
Crystal violet or gentian violet, also known as methyl violet 10B or hexamethyl pararosaniline chloride, is a triarylmethane dye used as a histological stain and in Gram's method of classifying bacteria. Crystal violet has antibacterial, antifungal, and anthelmintic properties and was formerly important as a topical antiseptic.The medical use of the dye has been largely superseded by more.

Polaire en apolaire moleculen Mr. Chadd Academy YouTube
Sommige stoffen zijn polair, andere apolair. Een molecuul is polair als de elektronen (die een negatieve lading hebben) in dat molecuul dusdanig verdeeld zij.

Invloed van apolaire staart op oplosbaarheid van alkanolen in water YouTube
If you look at the Lewis structure for HNO3 we can see that it is not a symmetrical molecule. We have Oxygen atoms on one side and a Hydrogen atom on the ot.

Synthese polair apolair YouTube
Learn to determine if PCl3 (Phosphorous trichloride) is polar or non-polar based on the Lewis Structure and the molecular geometry (shape).We start with the.

MkMolcules organiques
Aldrich-70862; 4-Amino-1-naphthalenesulfonic acid sodium salt hydrate technical; CAS No.: 123333-48-2; Synonyms: 1-Naphthylamine-4-sulfonic acid sodium salt; Naphthionic acid sodium salt; Sodium 4-amino-1-naphthalenesulfonate; Linear Formula: H2NC10H6SO3Na · xH2O

Polair apolair + begin chemisch rekenen filmpje groter YouTube
IUPAC Standard InChIKey: HZYHMHHBBBSGHB-ODYTWBPASA-N Copy CAS Registry Number: 557-48-2 Chemical structure: This structure is also available as a 2d Mol file.

Is een gegeven molecule polair of apolair? YouTube
Infobox references. [18F]Fluorodeoxyglucose ( INN ), or fluorodeoxyglucose F 18 ( USAN and USP ), also commonly called fluorodeoxyglucose and abbreviated [18F]FDG, 2- [18F]FDG or FDG, is a radiopharmaceutical, specifically a radiotracer, used in the medical imaging modality positron emission tomography (PET).

7 VWO Dipoolmomentdipolenpolairapolair scheikunde YouTube
Learn to determine if BCl3 is polar or nonpolar based on the Lewis Structure and the molecular geometry (shape).We start with the Lewis Structure and then us.
Huishoudelijke apparaten Silicagel polair of apolair
Begrippen polair en apolair. Verschuiving van bindingselektronen. Polaire of apolaire binding. Symmetrische of asymmetrische moleculen. Polair of apolair molecule.

Polaire en apolaire moleculen Tussentijdse toets Downloadbaar lesmateriaal KlasCement
Its chemical formula is CH3OH containing one carbon atom, one oxygen, and four hydrogen atoms. The molecular mass of methanol is 32.04 g per mol. It is calculated as. 1* (mol mass of C) + 4* (mol mass of H) + mol mass of O. 12+1×4+16 = 32 grams. Methanol does not have texture, it is a colorless liquid and smells like ethanol.

HAVO Hfst2 opgave 29 polair apolair YouTube
About Press Copyright Contact us Creators Advertise Developers Terms Privacy Policy & Safety How YouTube works Test new features NFL Sunday Ticket Press Copyright.

Oplossingen polair of apolair YouTube
Yes, caffeine is a polar molecule. The Oxygen and Nitrogen molecules have a stronger polarity than Carbon, allowing them to slightly pull the electrons towards them in their covalent bond. This will give those atoms a slightly negative charge while giving the Carbon a positive charge. This is the exact definition of polarity itself.

Chemie oplosbaarheid van stoffen (polair/apolair) YouTube
Yes, NH3 (Ammonia) molecule is polar in nature because of its asymmetrical shape ie; trigonal pyramidal structure, and the difference in electronegativities of N (3.04) and H (2.2). The charges over the nitrogen and hydrogen atoms are unequally distributed which results in a net dipole moment making NH3 (Ammonia) a polar molecule.

Polair & Apolair YouTube
The gas is made of two types of atoms - Carbon and Oxygen. Each CO2 molecule has two oxygen atoms and one carbon atom. If you study the CO2 Lewis structure and its molecular geometry, you will know that the Carbon atom is in the central position forming double bonds with both Oxygen atoms. This structure can help us understand the polarity of this molecule.